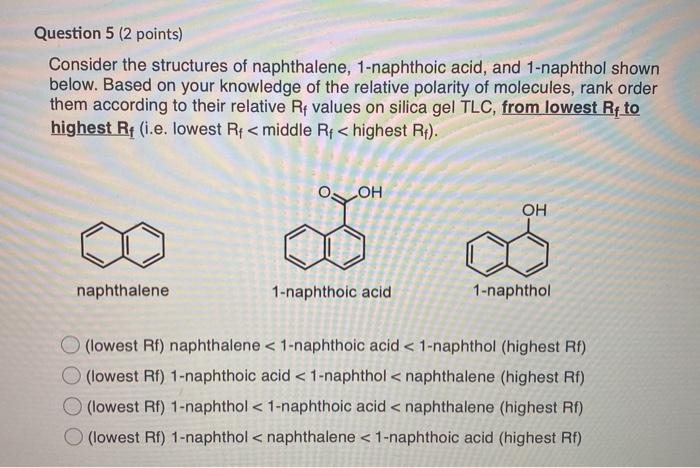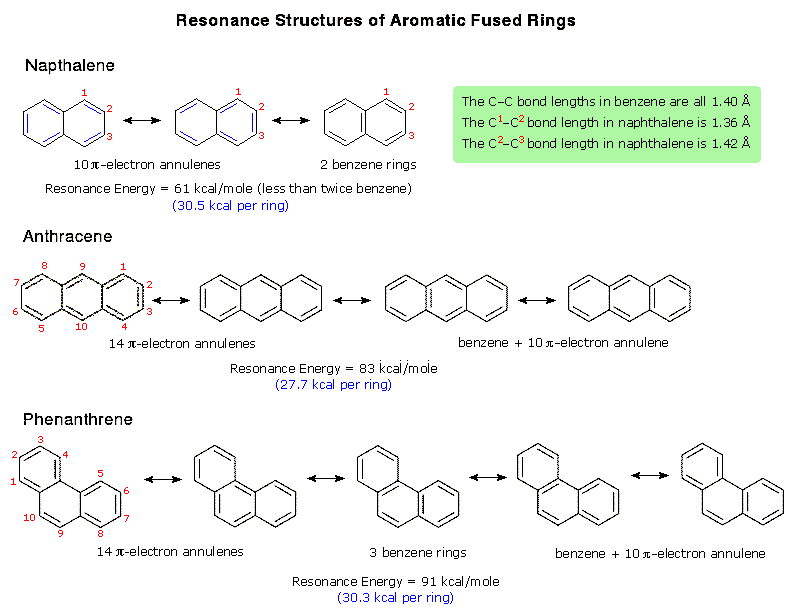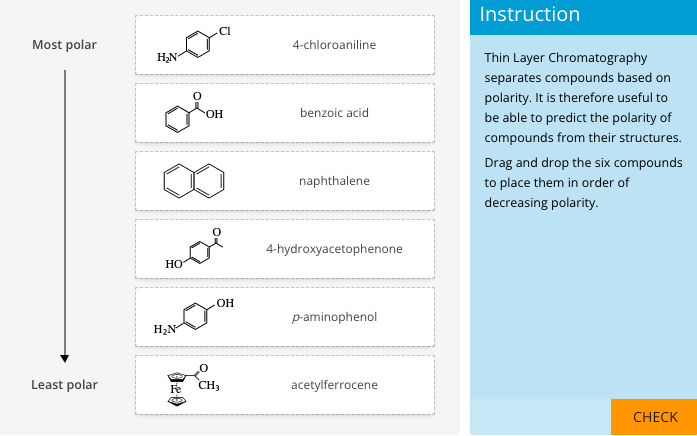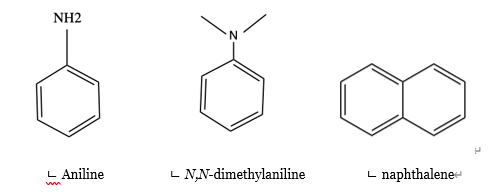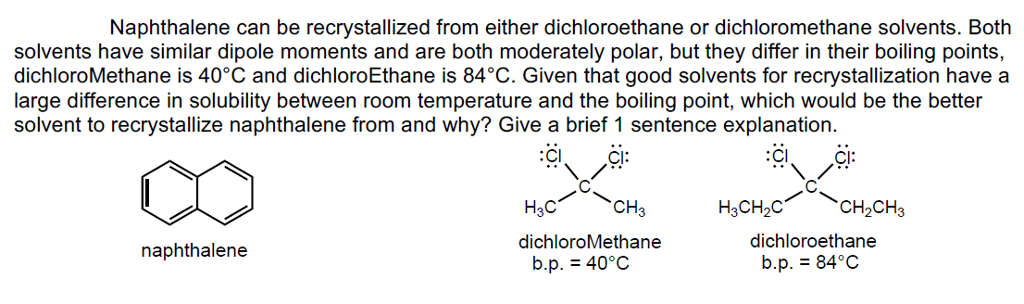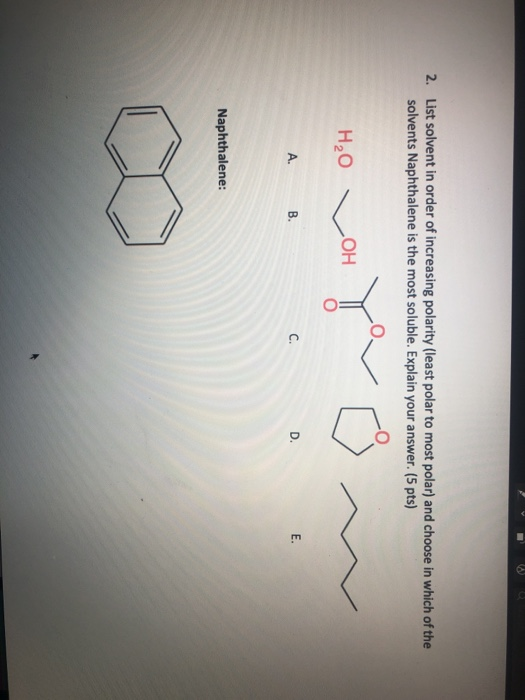
Introduction of polar groups on the naphthalene scaffold of molecular tongs inhibiting wild-type and mutated HIV-1 protease dimerization - MedChemComm (RSC Publishing)

NICS(1) values for naphthalene (1c) and azulene (2c), together with a... | Download Scientific Diagram
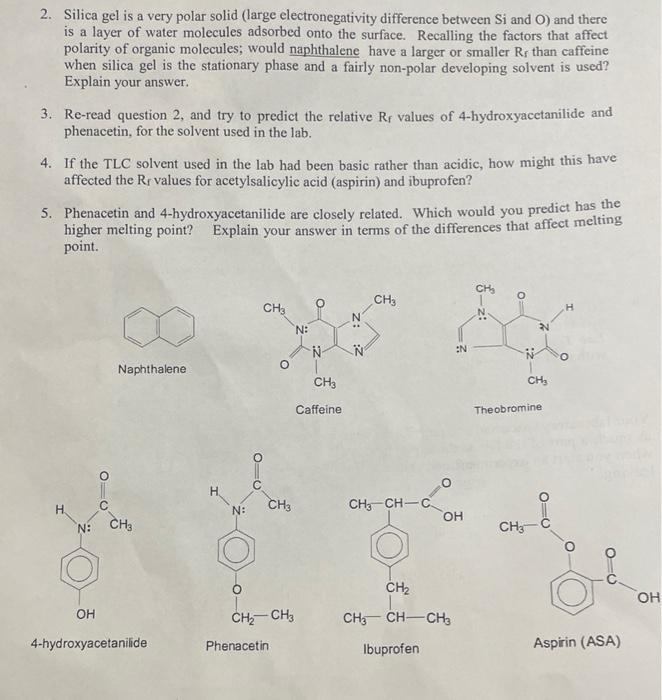
![PDF] Sorption of polar and nonpolar aromatic organic contaminants by plant cuticular materials: role of polarity and accessibility. | Semantic Scholar PDF] Sorption of polar and nonpolar aromatic organic contaminants by plant cuticular materials: role of polarity and accessibility. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/12dab10791507e62e8888347d8c6ce1d09dc9323/3-Table1-1.png)
PDF] Sorption of polar and nonpolar aromatic organic contaminants by plant cuticular materials: role of polarity and accessibility. | Semantic Scholar

Show the TLC when Naphthalene (a neutral unknown compound) is compared to the correct (or matching) standard and what the TLC would look like if Naphthalene (neutral unknown compound) was compared to

Valli took some common salt, naphthalene balls, camphor, baking soda and washing soda. She attempted to dissolve these substances either in water or in acetone. Complete the table with the expected results.

Table 1 from Fluorescence of 8-(Phenylamino)-1-naphthalene-ammonium- sulfonate in Solvents of Different Polarity | Semantic Scholar

Chromatogram for a mixture of benzene, naphthalene, and anthracene with... | Download Scientific Diagram

Chapter 14 Solutions | Organic Chemistry Plus Masteringchemistry With Etext -- Access Card Package 9th Edition | Chegg.com